
Also to keep in mind is the "leak-down" rate of the rings and valves, which also effects pressure readings. In addition, since most compression readings are taken with a warm or hot engine, the maximum compression pressures that are measured are hard to predict given that not all engines are measured at the same ambient air temperature, engine temperature, speed of engine cranking, etc.all of which can effect pressure. This is why, even humble VW beetles running compression ratios of 7.7:1 to 8.4:1 can have compression pressures in the 140-to-160 lbs./sq.in. This results in the gas increasing in both pressure AND temperature.resulting in a higher measured pressure than the diabatic case. That is, no heat enters or leaves the system. However, for the compression cycle that the Porsche (and just about every other normally aspirated, spark-ignited engine running on the Otto-cycle) has is that the compression is essentially ADIABATIC. for a 15:1 ratio the pressure would be abot 220 lbs/sq. Thus for a 10:1 compression ratio at sea level, the pressure would be 147 lbs/sq. What this means, if PV = nRT, is that pressure IS linearly inversely proportional to volume because the temperature increase that accompanyed the compression (because of the work of compression)is allowed to leave the system. The main point of your statement is true, but the calculation you show is for a compressed gas obeying (as I remember) Boyle's Law for a DIABATIC compression.that is, one where the temperature of the gas is the same at the beginning and end of the compression. 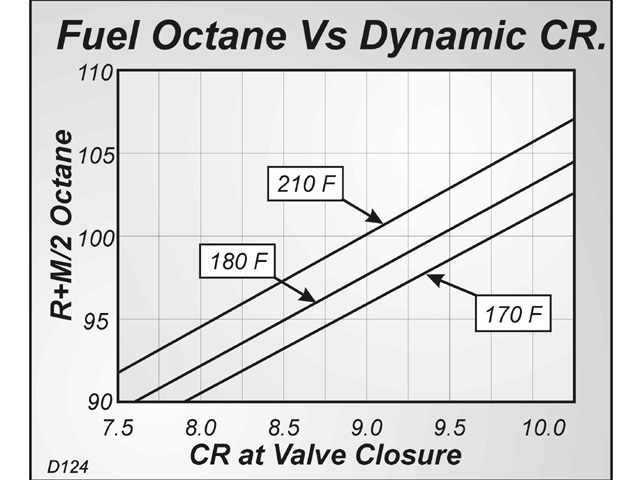
Just a long winded comment on your calculation of compression pressures from compression ratios (and vice-versa).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
